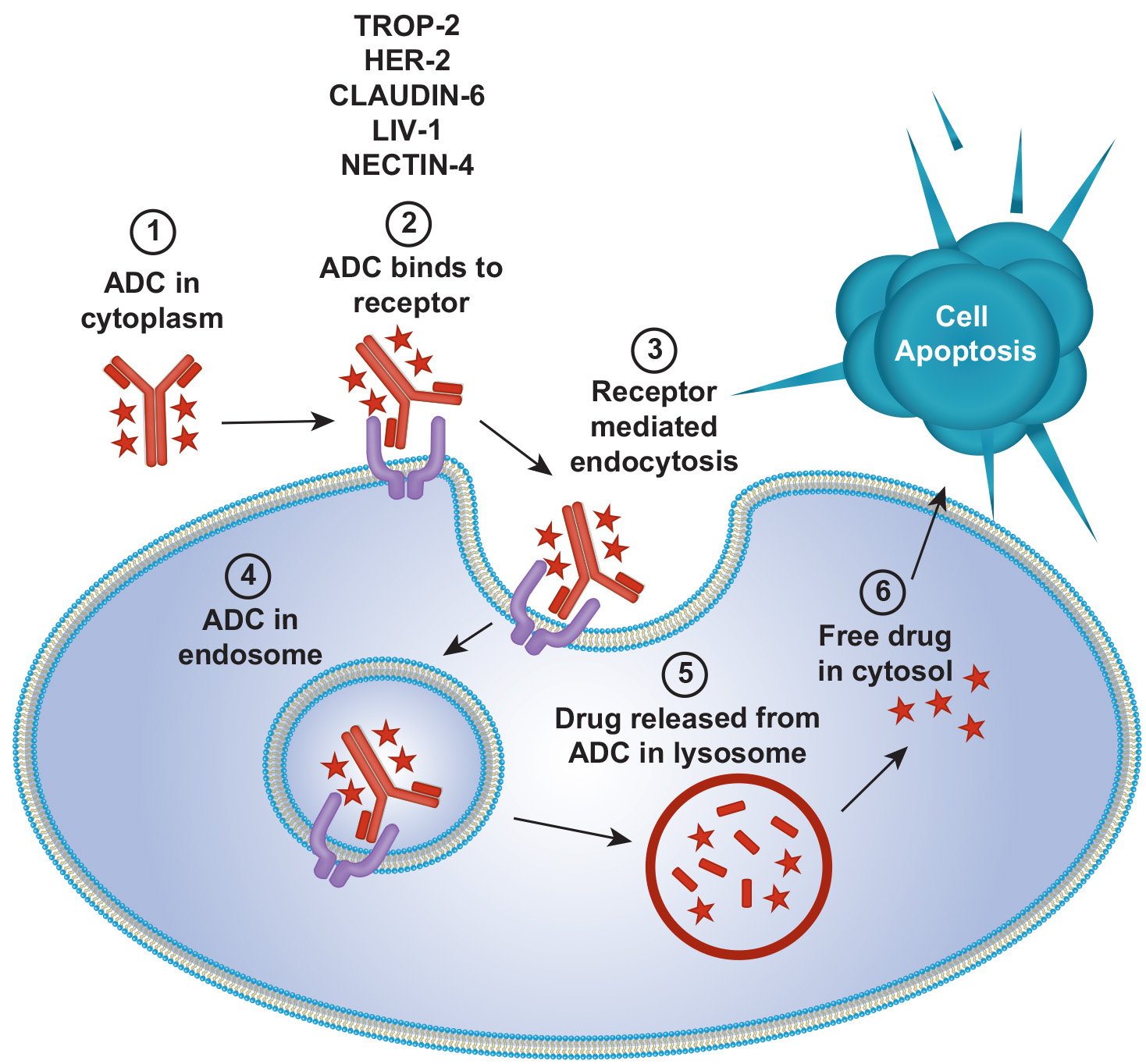
Antibody Drug Conjugates
Antibody-drug conjugates (ADCs) offer a refined approach to cancer therapy by targeting tumor cells with potent cytotoxic agents, minimizing harm to normal cells. They have become a standard treatment for breast cancer, effective across HER2+, HR+, and triple-negative subtypes.
ADC advancements have widened eligibility to patients with low or variable target antigen expression. Recent label expansions include Trastuzumab Deruxtecan to HR+, HER2‑low/ultralow after ≥1 ET in the metastatic setting.
Datopotamab deruxtecan (Dato‑DXd; Datroway) approved (Jan 17, 2025) for previously treated HR+/HER2‑ mBC after prior ET and chemotherapy.
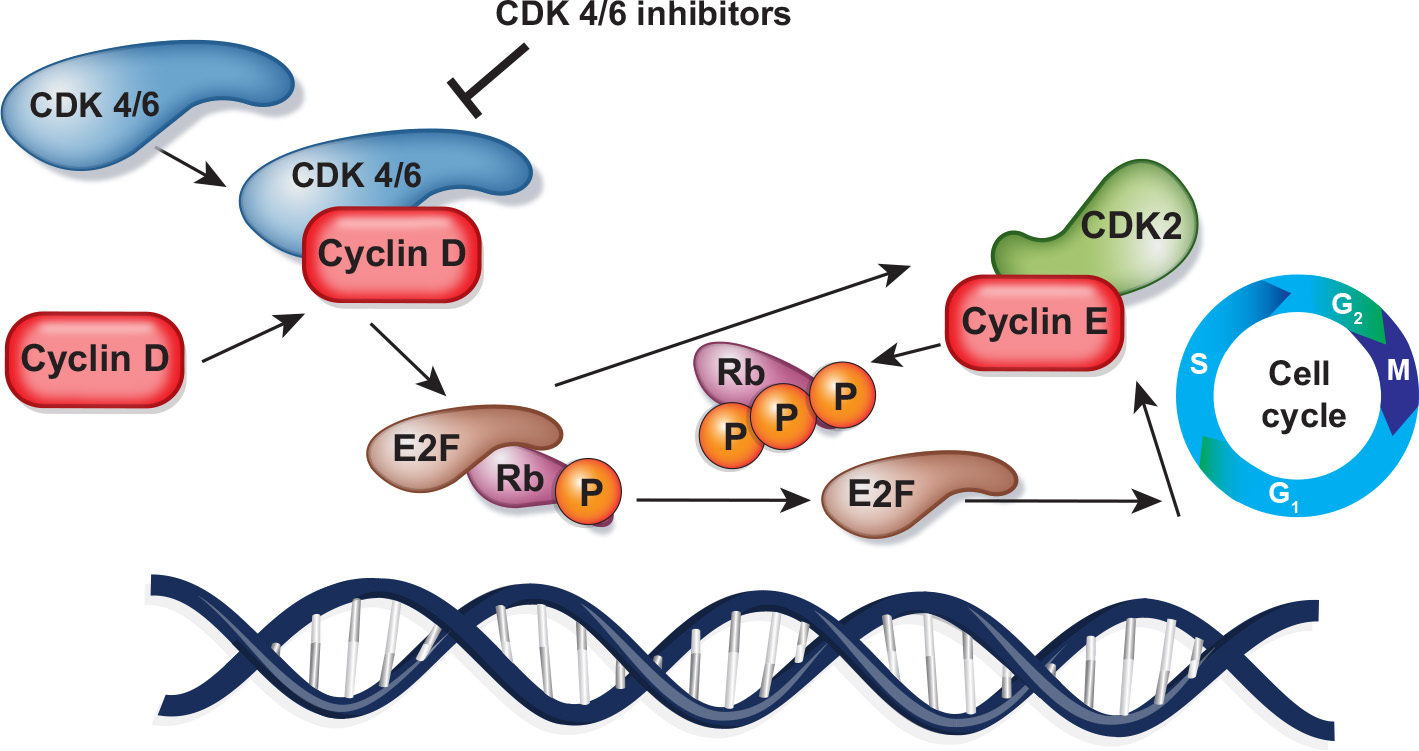
CDK inhibitors
Aberrant CDK4/6 pathway activation drives uncontrolled breast cancer cell proliferation.
Combining CDK4/6 inhibitors with endocrine therapy is a leading treatment for HR+/HER2- advanced breast cancer.
Next-generation CDK2 and CDK4 inhibitors, designed to be more specific and potent, may overcome resistance to first-generation CDK4/6 inhibitors and broaden the use of CDK inhibitors beyond ER+ breast cancer.
Ribociclib approved (Sept 17, 2024) with an aromatase inhibitor for high‑risk stage II–III HR+/HER2‑ early breast cancer (NATALEE).
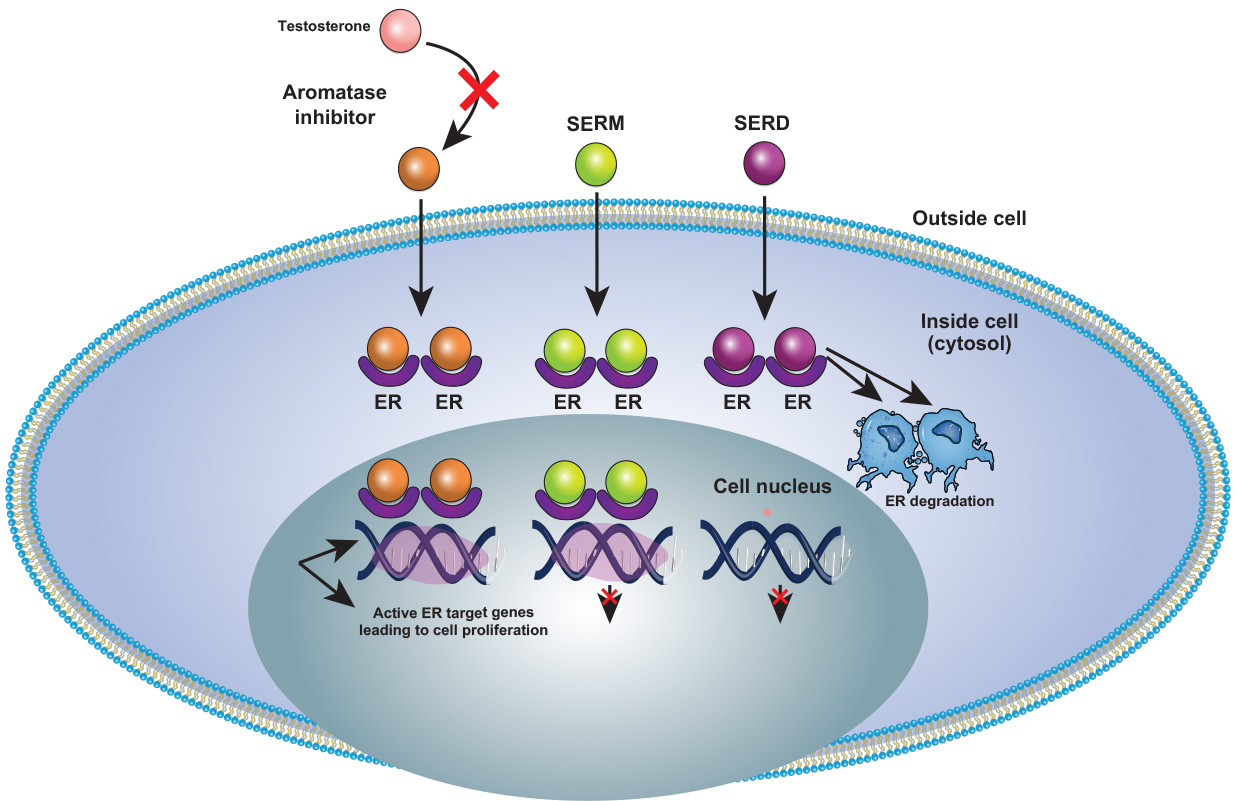
ER Degraders/Modulators
Anti-estrogen therapy targeting the estrogen-mediated signaling pathway is an essential component of treatment for both early and advanced-stage breast cancer expressing the estrogen receptor (ER) and/or progesterone receptor (PR).
For patients with ER-positive, HER2-negative breast cancer, Fulvestrant (SERD) has been the standard of care after progression on CDK4/6 inhibitors and endocrine therapy.
FDA approved Elacestrant (Orserdu) in patients with ESR1 mutations based on findings from the phase 3 EMERALD trial.
New next-generation hormonal agents (SERD, CERAN, and PROTACs) promise to transform treatment, offering oral alternatives that reduce the need for invasive delivery and address acquired variants like ESR1.
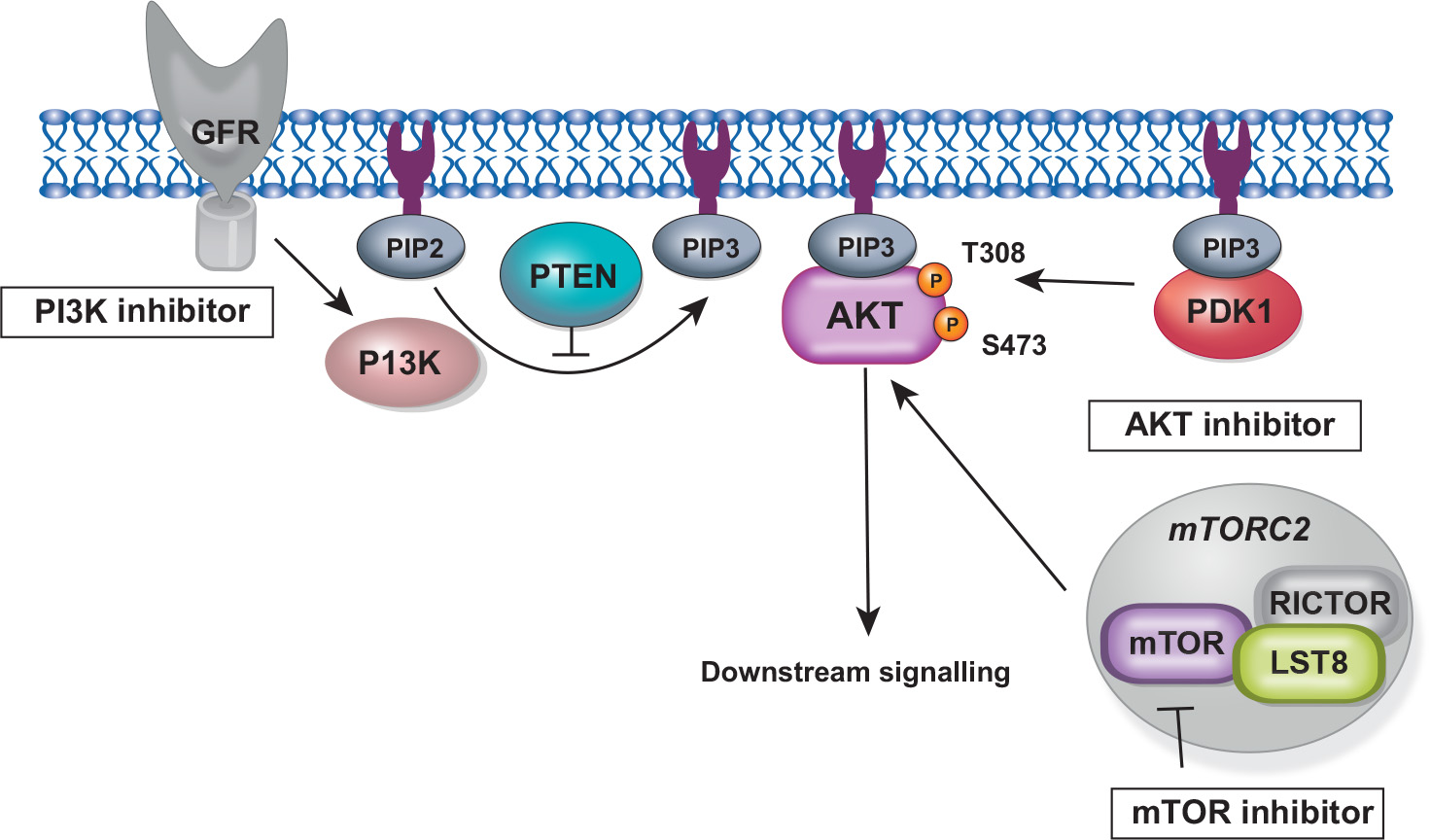
PI3K/AKT/mTOR
The occurrence and development of breast cancer are driven by gene mutation and dysregulation of cell signaling pathways, and PI3K–AKT signaling is the most upregulated pathway in breast cancer.
The PI3K/AKT/mTOR signaling is over-activated in about 70% of breast cancers and is altered in 35% of primary TNBC. This pathway’s dysregulation contributes to endocrine and anti-HER2 resistance and is more frequent in HR+ breast cancer.
Recently, Inavolisib (PI3K‑α; Itovebi) + palbociclib + fulvestrant got approved on Oct 10, 2024 for endocrine‑resistant, PIK3CA‑mutated HR+/HER2‑ advanced disease.
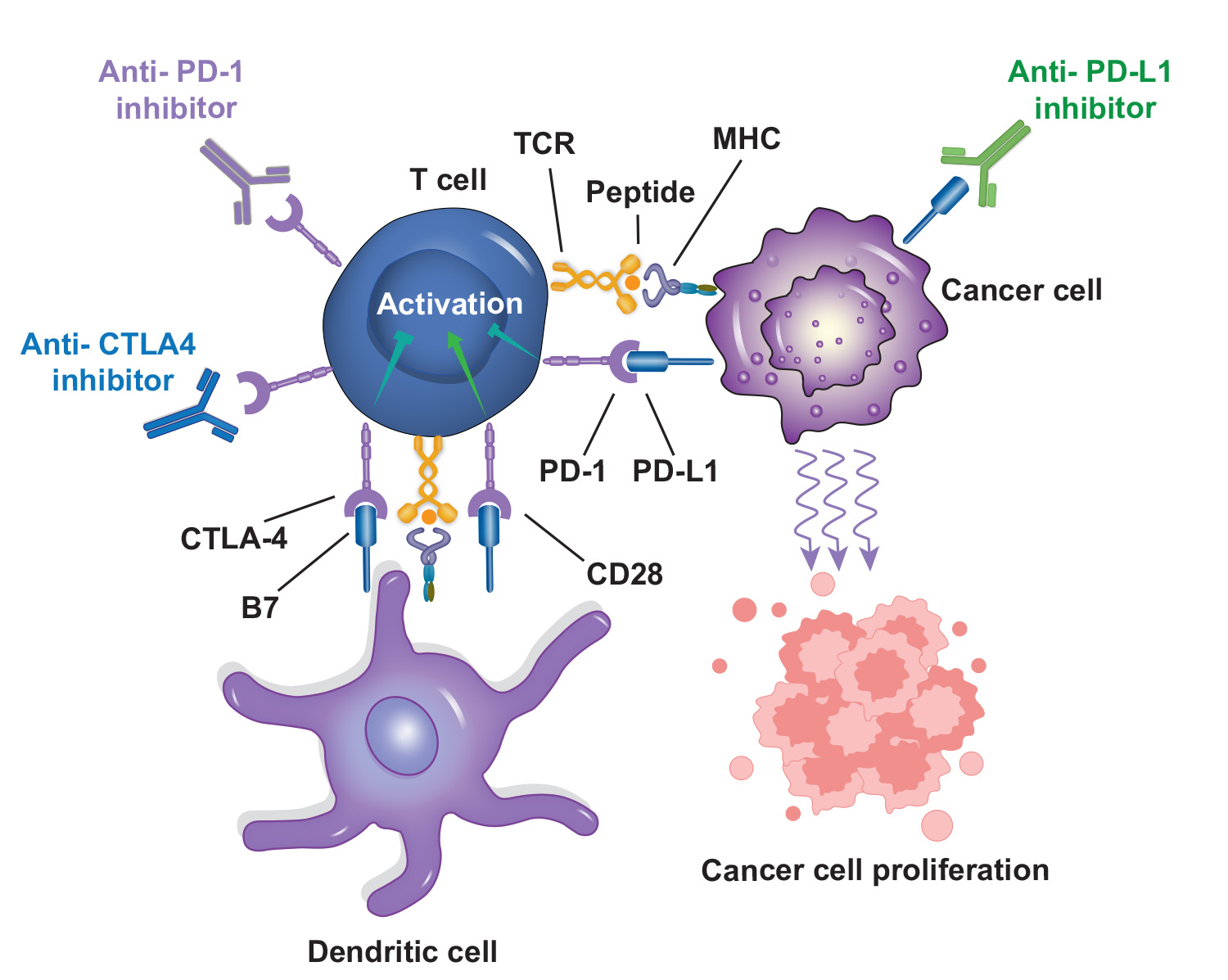
Immunotherapy in Breast Cancer
Immunotherapy, alone or with chemotherapy, has shown clinical value in various cancers, including TNBC by improving PFS and OS. However, immune checkpoint inhibitors are associated with unique immune-related adverse events, distinct from the side effects of standard chemotherapy.
The most studied co-inhibitory receptor is programmed death-1 (PD-1). Nivolumab and pembrolizumab are the primary clinically approved PD-1 inhibitors and have vastly altered the treatment of many advanced, metastatic and unresectable cancer types.
Novel inhibitory immune checkpoint proteins being investigated in early-stage clinical trials in a breast cancer setting include TIM-3, VISTA, and B7-H3. Immune checkpoint targets, such as CTLA-4, LAG3, and TIGIT, aim to prevent T cell inhibition, while molecules like OX-40 and 4-1BB stimulate T cells to enhance their cytotoxic activity.
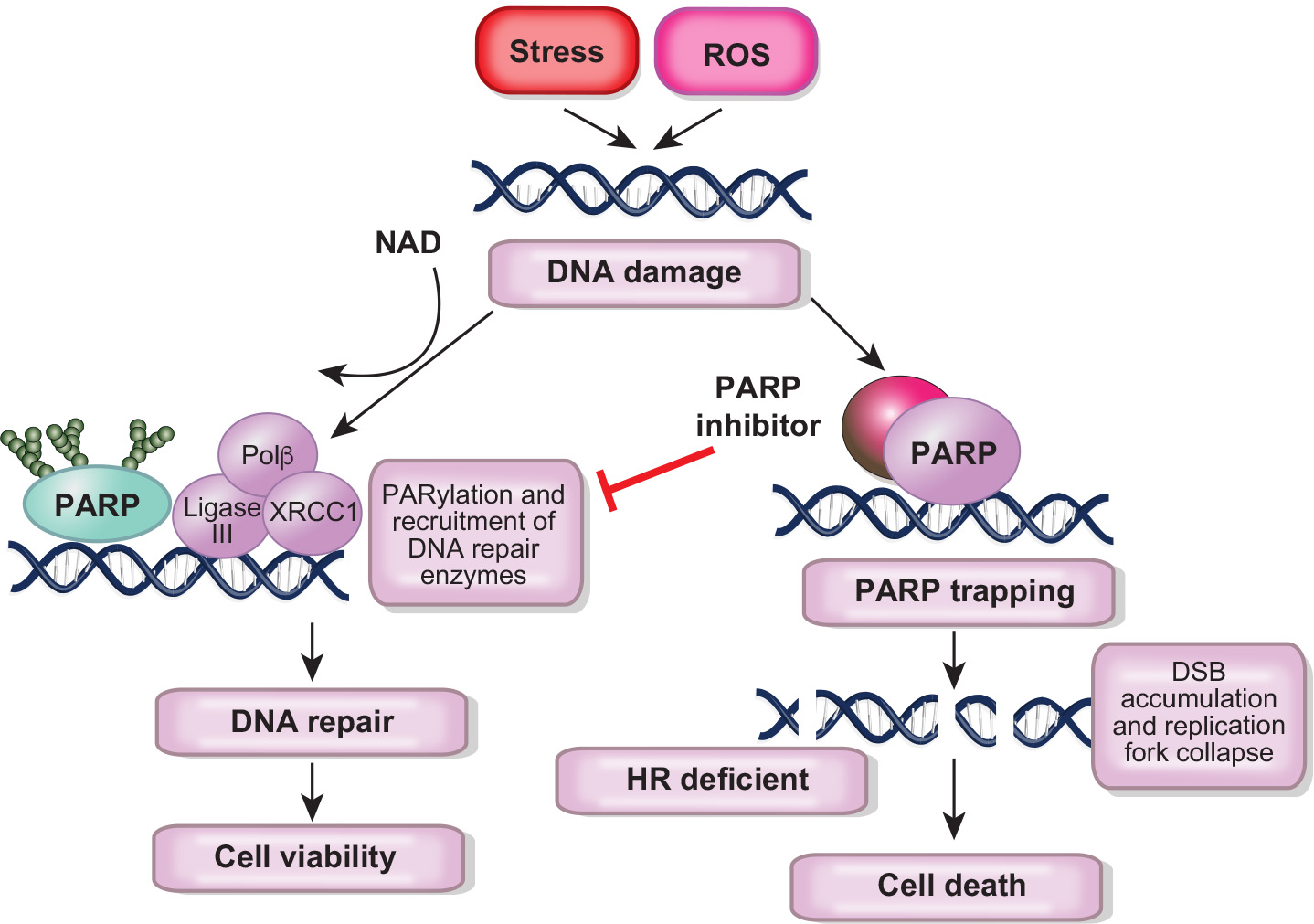
PARP inhibitors in Breast Cancer
PARP (Poly ADP-ribose polymerase) inhibitors have emerged as a promising therapeutic intervention in breast cancer, particularly in germline BRCA mutation (gBRCAm) carriers.
In breast cancer, two PARP inhibitors, Olaparib and Talazoparib, have been approved for treatment of gBRCAm carriers with metastatic HER2-negative breast cancer based on the OlympiAD and EMBRACA trials, respectively.
Despite the early and dramatic response seen with PARP inhibitors, patients receiving them often develop treatment resistance.